Abstract
Objective
The purpose of this large, population-based study was to investigate preoperative comorbidities as risk factors of mortality in pediatric liver transplant recipients.
Methods
A total of 2,938 patients who underwent liver transplantation (LT) surgery from 1998 through 2012 in Taiwan were enrolled in this study. Based on the International Classification of Disease, 9th Revision, Clinical Modifi cation (ICD-9-CM) codes, basic information regarding medical comorbidities was extracted from the National Health Insurance Research Database (NHIRD).
Results
All patients were followed to the endpoint of the study or until death. The study enrolled 2,597 adult (≥ 18 years old) and 341 pediatric (< 18 years old) liver transplant recipients. The median age for the pediatric cohort was 1.88 years (interquartile range = 0.92–5.42 years). Four hundred and twenty-eight deaths occurred after LT in the total population, including 41 children. The median follow-up period was 6.1 years (interquartile range = 2.5–9.7 years) in pediatric liver transplant recipients. Pediatric patients with heart disease exhibited the highest risk of mortality. Further, during the entire study period of 14.5 years, patient survival rates were signifi cantly different (log-rank p = 0.002) for patients younger than 18 years and those older than 18 years.
Conclusion
Cardiac disease is an important risk of mortality in pediatric LT. These fi ndings confi rm that the survival rate of LT is higher in pediatric patients than in adult patients.
Keywords
pediatric recipients ; liver transplantation ; mortality ; population-based study ; cohort study
Introduction
The successful experimentation in canines paved the road for the first attempt of orthotopic liver replacement in humans in 1963. As an index of history, we cannot ignore one important person, Thomas E. Starzl.1 Although the surgery was not successful and the patient died, he still created a landmark journey for pediatric liver transplantation (LT). His pioneer contribution has become a routinely successful clinical procedure. Initially, the mortality rate was not good because of a high rate of organ rejection. Since the development of immunosuppressants, organ rejection rates have significantly improved, and survival rates have significantly increased.
Since 1983, LT has become the standard treatment for pediatric end-stage liver disease (PELD). Because of its difficult procedure and the size limitations of donor liver, the survival rate was quite low. The survival rate greatly increased in the late 1980s through the 1990s because of the advancement in surgical techniques, such as split cadaveric LT, reduction in the size of adult liver donation and living donor LT.2,3 LT has become a very successful treatment for children with end-stage liver disease and provides a chance of long-term, healthy survival.
Some studies have indicated that the most common cause of death in pediatric liver transplant patients was graft failure.4 The age and weight of both, the liver transplant donor and recipient were also identified as risk factors for mortality and morbidity.5 However, the effects of initial pre-liver transplant comorbidities have seldom been considered. In order to evaluate the pre-liver transplant comorbidities as risk factors for mortality, we retrospectively reviewed all liver transplants performed between 1998 and 2012 that were recorded in Taiwan’s National Health Insurance Research Database (NHIRD).
Methods
Study Design
This retrospective observational cohort study was performed based on information from a registered national database. We collected data on LT recipients from the NHIRD, a universal insurance program that started in March 1995, which was reformed from 13 previous health schemes.
Fig. 1 shows the flow chart for patient identification and selection. Data from the NHIRD were collected from the time health insurance began to pay for LT in 1998 until 2012 by using the International Classification of Disease, 9th Revision, Clinical Modification (ICD-9-CM) codes V427 (LT status) and 996.82 (complications of transplanted liver). During this period, a total of 4,086 LT recipients were recorded in this large database. Due to the lack of the LT surgery codes (505, 75020A, or 75020B) between 1998 and 2012, 1,148 patients were excluded from the study. Therefore, the final number of the LT recipients available for analysis was 2,938. By classifying all LT recipients below the age of 18 year as the pediatrics group, the study cohort consisted of 341 pediatric and 2,597 adult LT recipients.
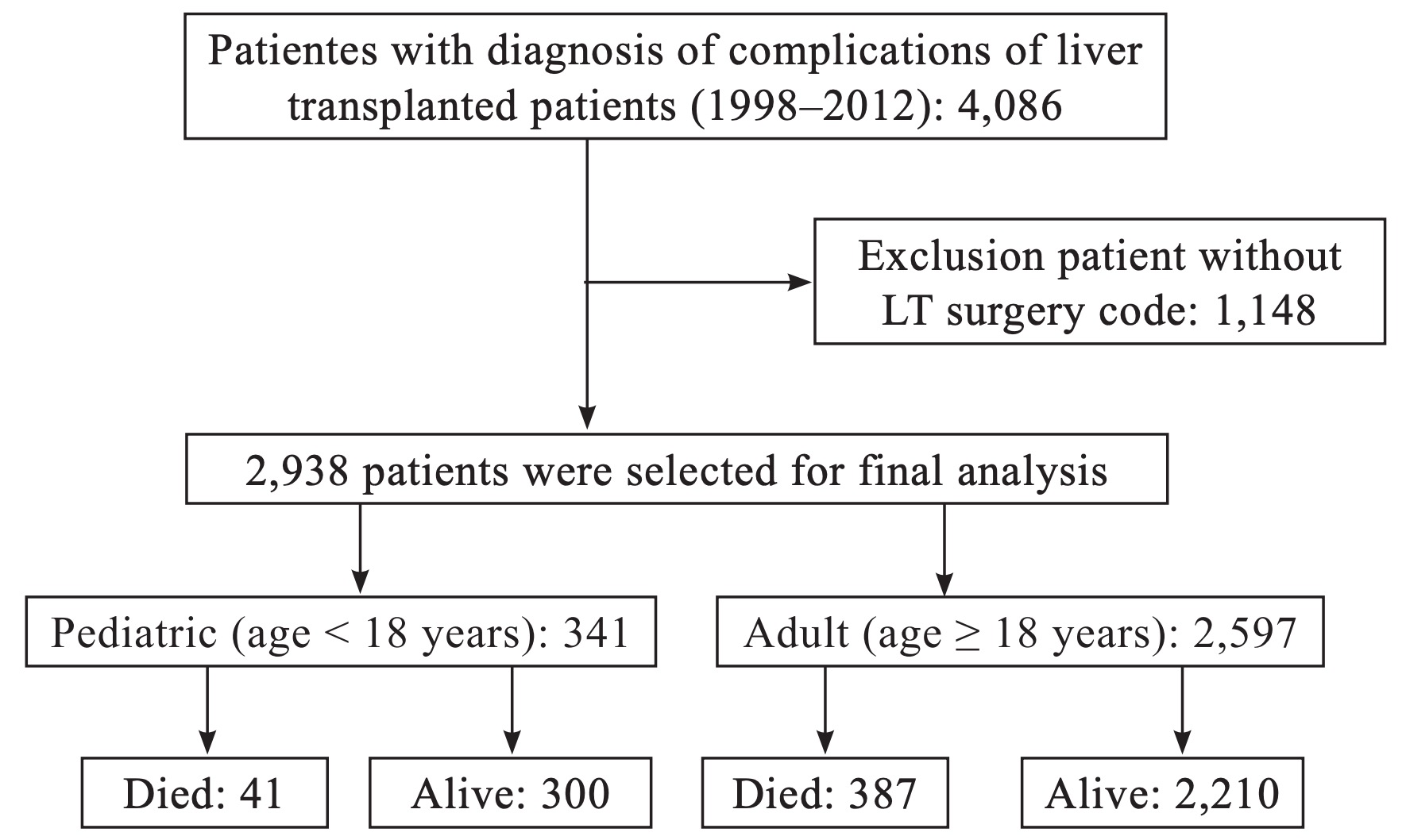
Download full-size image
LT: liver transplantation.
Data Source
From the NHIRD, all of Taiwan’s National Health Service hospital management warehouse data including admission could be obtained. The NHIRD contains data of 99.44% of the entire Taiwanese population (23.28 million). Most medical institutions (93.00%) have been contracted to the Bureau of National Health Insurance (NHI). The Bureau of NHI integration organized the claims data throughout the NHI system, in order to provide de-identified and computerized data, and established the NHIRD. The database contains basic information in addition to raw claims data, hospital medical claims, and clinical diagnostic codes according to the ICD-9-CM. According to the NHI, patients who need an LT should be diagnosed by a liver transplant surgeon or a gastroenterologist.
This study was evaluated and approved by the NHIRD Research Council (NHIRD-103-103) and Chang Gung Memorial Hospital (CGMH-103-0102B).
Study Variables
The demographics data of every LT procedure in Taiwan, including the time of transplant, age, sex, and medical comorbidities, were extracted. The following comorbidities, including hypertension, pulmonary disease, diabetes mellitus, cerebrovascular diseases, heart disease, renal failure, dialysis, cancer liver cirrhosis, chronic hepatitis, peptic ulcer, psychosis, esophageal varices, biliary atresia, Wilson’s disease, glycogen storage disease, and urea cycle metabolism disorders, were identified among patients in our study cohort. Preoperative medical records in outpatient or inpatient diagnosis were verified using ICD-9-CM diagnosis codes.
Statistical Analysis
The primary study outcome was pediatric postLT mortality. SAS statistical software (version 9.3; SAS Institute Inc., Cary, NC, USA) was utilized for the data analysis. Descriptive statistics were used to estimate the frequencies. Categorical variables are presented as numbers (%) and continuous variables as means (± standard deviation) or medians (± interquartile range), dependent upon the normality of distribution. Differences between groups were assessed using the chi-square test or Fisher’s exact test for categorical variables. Risk ratios were used to compare the pediatric LT recipients to the adult LT recipients and the pediatric LT recipients who survived to those who died. Kaplan–Meier estimates with log-rank tests were used to compare the survival during follow-up among the age cohorts. In the mortality analyses, patients were followed up until death or to the end of the follow-up period, whichever occurred first; p values < 0.05 and < 0.001 were considered significant and highly significant, respectively.
Results
The data of 4,086 LT recipients in Taiwan were present in the NHIRD. After excluding those who lacked a code for LT surgery during the study period (n = 1,148), the cohort consisted of 2,938 participants. In addition, the participants were classified as adult (n = 2,597, 88.4%) or pediatric (n = 341, 11.6%) LT recipients for further analysis.
Pediatric Cohort
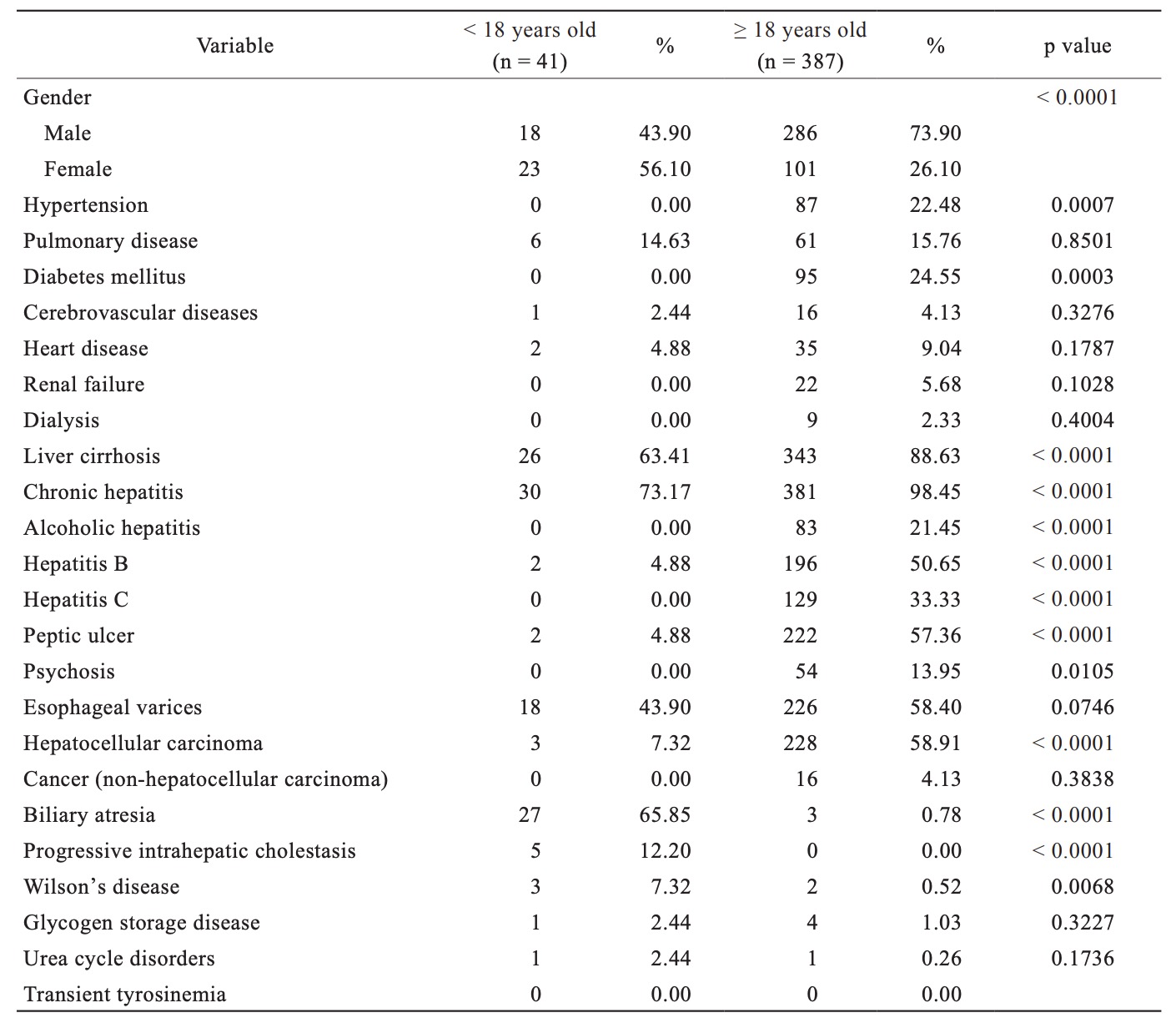
Table 1 highlights the baseline demographic differences between the pediatric and adult LT cohorts. As expected, pediatric LT recipients were less likely to have any medical comorbidity. Adult patients were more likely to have hypertension, pulmonary disease, diabetes mellitus, cerebrovascular diseases, heart disease, renal failure, liver cirrhosis, chronic hepatitis, peptic ulcer, psychosis, esophageal varices, and hepatocellular cancer than pediatric patients. Pediatric patients were more likely to have congenital diseases, such as biliary atresia, progressive intrahepatic cholestasis, and Wilson’s disease. Moreover, chronic hepatitis was the most common medical comorbidity in both groups.
Download full-size image
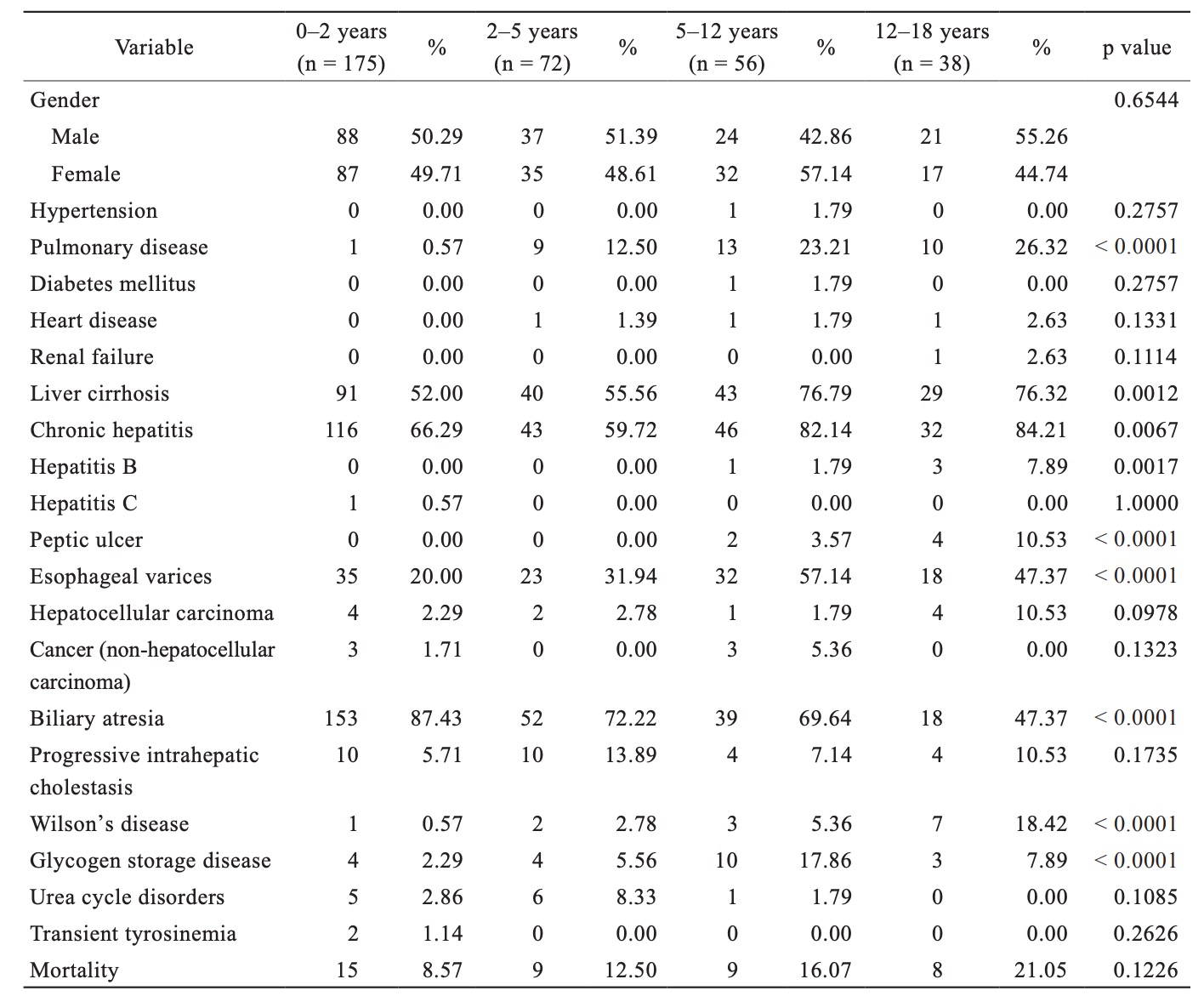
Table 2 illustrates a comparison of baseline demographics of pediatric LT recipients among various age groups in Taiwan between 1998 and 2012. The median age for the pediatric cohort was 1.88 years (interquartile range = 0.92–5.42 years), and the male:female sex ratio percentage was 49.9:50.1. The most common indication for LT in pediatric patients was biliary atresia (n = 262, 76.83%), especially in infant patients (age < 2 years). Chronic hepatitis and liver cirrhosis were the most common medical comorbidities in pediatric LT recipients. There was no significant difference in survival rates within the pediatric cohort during a median follow-up period of 6.1 years: age 0–2 years (95.5%), age 2–5 years (86.6%), age 5–12 years (83.0%), and age 12–18 years (77.2%), respectively (log-rank p=0.12).
Download full-size image
Post-LT Mortality
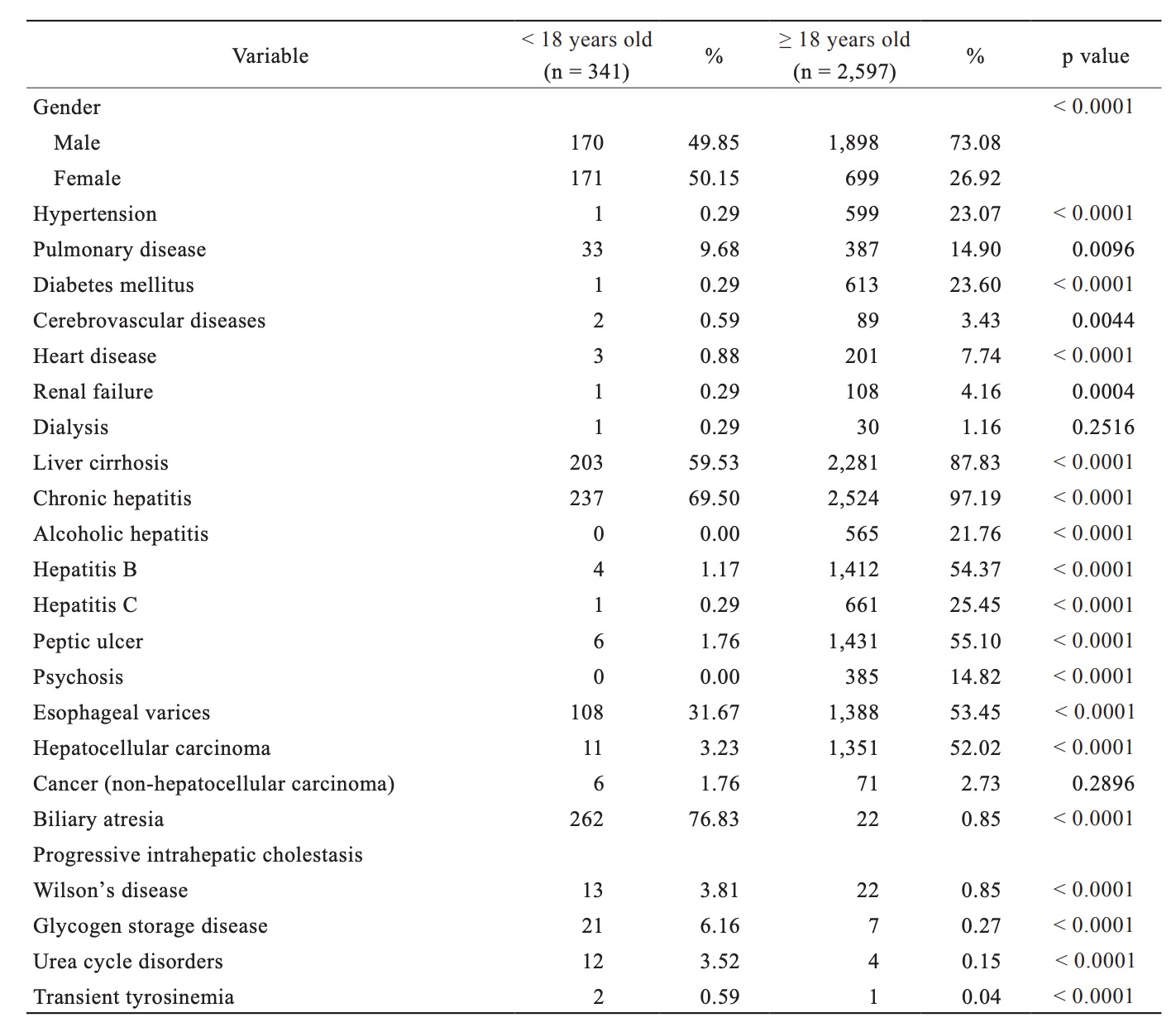
We identified a total of 428 deaths, which occurred post-LT in Taiwan over the past 14.5 years. Of the 41 pediatric deaths, 23 occurred within the first year post-LT and 18 occurred beyond the first year. The median follow-up period was 6.1 years (interquartile range = 2.5–9.7 years) in pediatric LT patients. Table 3 highlights baseline demographic differences between the pediatric and adult LT cohorts who died post-LT. Adult patients who died post-LT were more likely to have hypertension, diabetes mellitus, liver cirrhosis, peptic ulcer, and hepatocellular cancer.
Download full-size image
In Table 4, we compared baseline demographics between the pediatric LT recipients that were still alive and the patients that deceased after transplantation. Pediatric LT recipients with heart disease constituted a higher proportion of the cohort that died in comparison to non-heart disease recipients, but no other significant differences were observed between pediatric LT recipients who were alive vs. deceased patients.
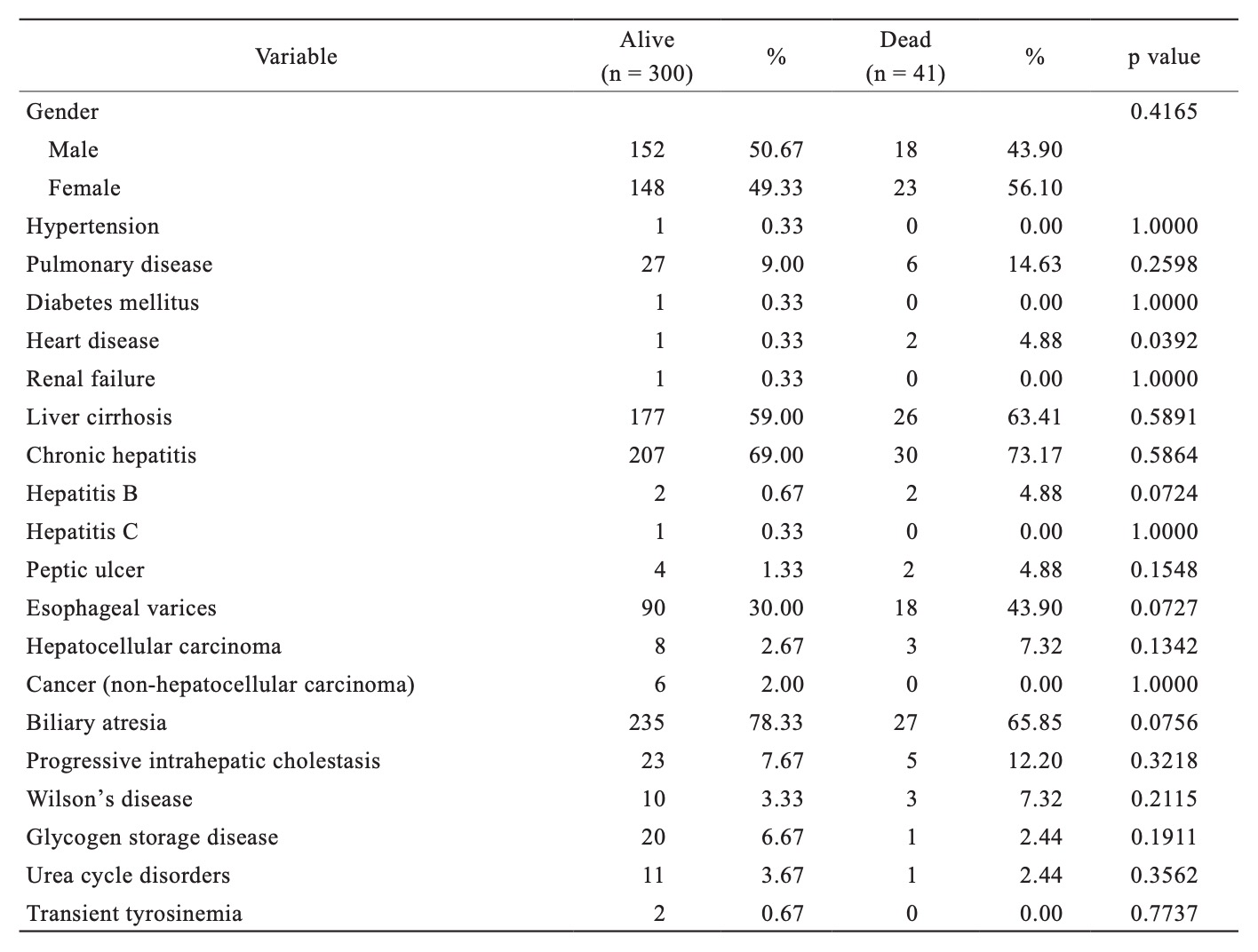
Download full-size image
This study identified 342 deaths that occurred post-transplantation during a median follow-up period of 2.85 years (interquartile range = 1.2–5.5 years) of all LT patients. Fig. 2 presents the unadjusted Kaplan– Meier survival curves for pediatric patient survival according to recipient age after LT during the 14.5- year study period. Neither first-year patient survival between four age groups < 18 years, nor the overall 14.5-year patient survival rates in these 4 groups were significantly different (log-rank p=0.06 and 0.12, respectively). Fig. 3 presents the unadjusted Kaplan– Meier survival curves between pediatric patients (< 18 years) and adult patients (≥ 18 years) after LT during the 14.5-year study period. During the first five years, patient survival rates were similar between the < 18 years and ≥ 18 years groups, showing no significant difference (log-rank p=0.06). However, during the entire study period of 14.5 years, patient survival rates between the two groups were significantly different (log-rank p=0.002). These findings indicate that the outcome of LTis better in pediatric patients than in adult patients during a long-term follow-up period.
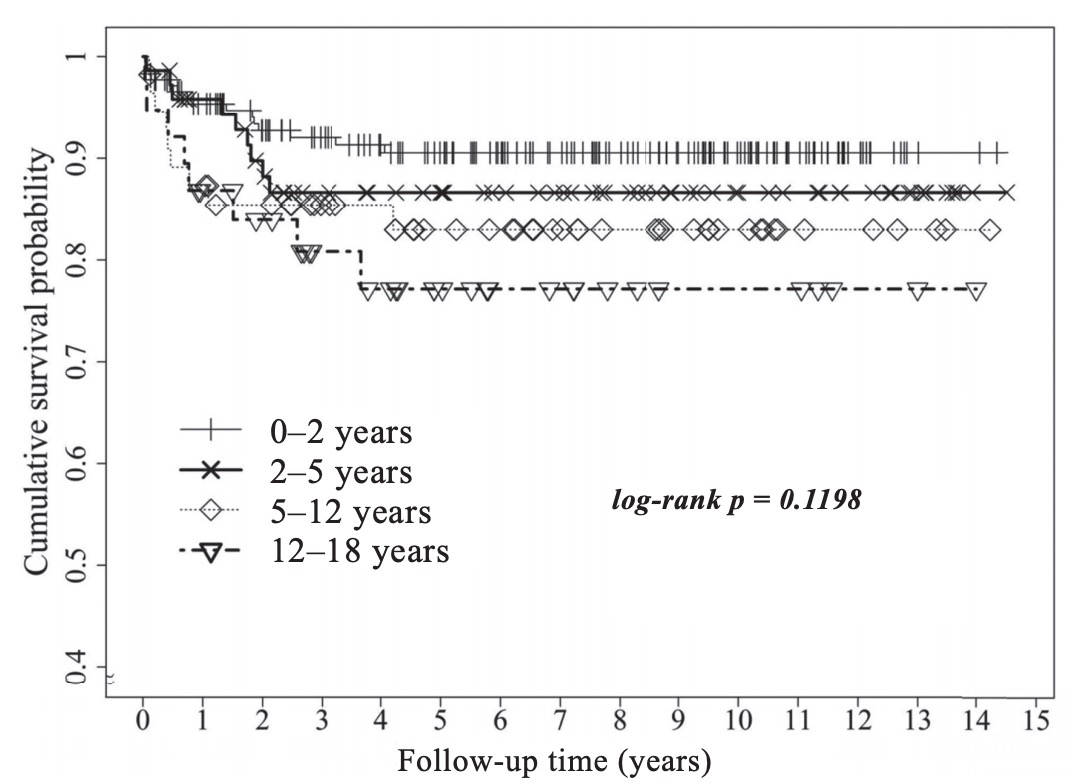
Download full-size image
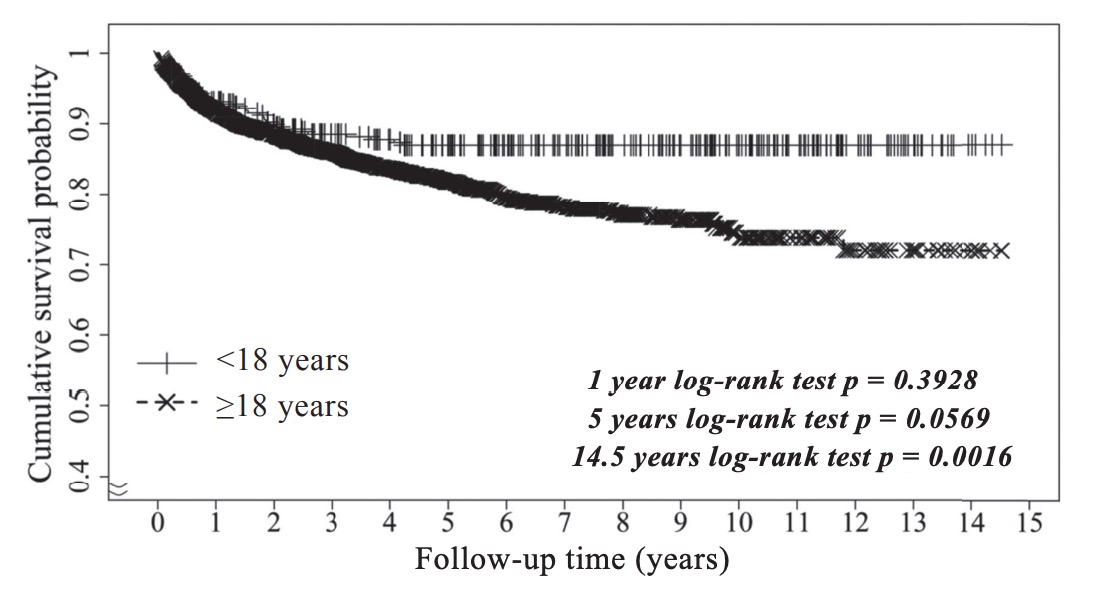
Download full-size image
Discussion
Consideration of pre-liver transplant medical comorbidities and clinical outcomes related to the transplant along with developing a relevant organ transplant allocation policy is essential. In addition, there are some concerns for pediatric LT recipients. To our knowledge, this is the first study from a large database that determines whether pediatric LT recipients have a significantly lower overall mortality than adult LT recipients.
Based on this observational cohort study over the past 1.5 decades in Taiwan, pediatric LT recipients had a mortality rate of 14.43% (median follow-up of 6.1 years after transplantation). Although there are important baseline differences between all pediatric and adult liver allograft recipients, there was surprisingly little to distinguish the pediatric vs. adult mortality cohorts post-transplantation during the first 5 years. Finally, only heart disease was associated with risk of pediatric-related mortality after transplantation, showing a slightly significant difference. Minor heart defects do not significantly affect the risk of pediatric LT.6-8 However, serious congenital heart diseases, especially right ventricular hypertrophy, might be the contraindication of pediatric LT.9 This is due to the higher risk of air embolism, hypoxia, and progressive heart failure, as well as the burden of hemodynamic change during LT. In order to provide and prioritize treatments in the best interest of patients, multidisciplinary discussions between transplant surgeons, pediatric cardiologists, and anesthetists are needed. Pulmonary diseases, which mostly may consider as the complication of severe cirrhosis, was one of the poor prognosis factor in adult LT, not in pediatric group.10 Proper treatments for children during and after LT, by using inhaled nitric oxide might improve hypoxia.11 In addition, chronic hepatitis12,13 and renal failure14-16 were considered as “post-LT” complications. However, our study showed that there were no statistically significant differences between the alive and the dead groups in pediatric LT recipients. Besides, due to the relatively small sample size in pediatric LT, further clinical prospective research will be recommended.
Obviously, when compared to adult liver allograft recipients, the survival rate in pediatric liver transplant recipients is better. Not surprisingly, this may be due to fewer preoperative medical comorbidities in children. We noticed that the incidence of chronic hepatitis in adults (97.19%) was greater than in pediatric (69.50%) LT recipients. In particular, individuals below the age of 18 years may have a lower risk of hepatitis B virus (HBV) infection, which can lead to chronic hepatitis and consequently liver cirrhosis,12 due to the Taiwanese government implementing the hepatitis B vaccine since 1984.13 The relationship between age-related comorbidities and postoperative mortality risk remains a concern to LT recipients.14,15 Preoperative evaluation and long term care in this particular transplantation group have unique challenges.14
Biliary atresia is the most common indication in pediatric LT recipients.16 Thus, from the beginning of the pretransplantation workup process, nutritional status should be maximized, which influences the results either before or after the transplantation, particularly in the pediatric population. Biliary atresia increases the incidence of cholestatic liver disease, which leads to fat malabsorption, causing a deficiency of calories as well as fat-soluble vitamins.16 Optimizing the nutritional status of LT children has translated into improved survival rates of pediatric LT patients, lower incidence of infections, and fewer surgical complications.17
Limits of the Study
The statistical power of this population-based study was sufficient to delineate pediatric preoperative comorbidities as risk factors of mortality or not. However, there are still some limitations that need to be disclosed.
First, the NHIRD is a large secondary reimbursement claims database that lacks details about the information of patients’ physical examinations, laboratory data, and PELD scores,18,19 which influence the mortality and the severity of pre-LT conditions.
Second, in this study, we used medical records and ICD-9-CM codes to define LT cases, pre-transplant medical comorbidities and mortality. Although the Bureau of NHI is responsible for regularly crosschecking samples of each hospital’s claims with the medical charts, miscoding due to human error is still possible by the clinical physicians.20 Recent studies have shown the accuracy of the diagnoses in the NHIRD main coding. However, validity of complications and comorbidity codes may still be one of the limitations of this study.21
Finally, although population-based health care management data have been used in LT- related research,22-24 the cause of death and allograft loss were not analyzed in our study. The causality of the correlation between comorbidities, complications, and LT mortality cannot be concluded and need to further prospective studies. This is because the causality of the correlation between comorbidities, complications, and LT mortality cannot be concluded without further prospective studies.
Conclusion
Liver transplant recipients below the age of 18 years show lower mortality rates compared to adult LT recipients during a long-term follow-up period. The difference in mortality between the two groups seems to be associated with fewer medical comorbidities. We believe that our findings should enable clinicians to perform better risk stratification for pediatric transplant candidates. We also recommend that further prospective studies be conducted to understand how age and comorbidities affect mortality in pediatric LT recipients.
Acknowledgments
This study was based in part on data from the NHIRD provided by the Bureau of NHI, Department of Health, and managed by the National Health Research Institutes. The interpretation and conclusions contained herein do not represent the views of the Bureau of NHI, Department of Health or National Health Research Institutes. This work was partially supported by grants from the Chang Gung Memorial Hospital (CORPG3E0131 and CORPG3E0132) to Huang-Ping Yu.
Conflicts of Interest
The authors report no conflicts of interest in this work.
References
| 1 |
Starzl TE, Marchioro TL, Porter KA, Brettschneider L.
Homotransplantation of the liver.
Transplantation 1967;5:790–803.
|
| 2 |
Broelsch CE, Emond JC, Thistlethwaite JR, Rouch DA, Whitington PF, Lichtor JL.
Liver transplantation with reduced-size donor organs.
Transplantation 1988;45:519–523.
|
| 3 |
Emond JC, Whitington PF, Thistlethwaite JR, Alonso EM, Broelsch CE.
Reduced-size orthotopic liver transplantation: use in the management of children with chronic liver disease.
Hepatology 1989;10:867–872.
|
| 4 |
Kelly D, Verkade HJ, Rajanayagam J, McKiernan P, Mazariegos G, Hübscher S.
Late graft hepatitis and fibrosis in pediatric liver allograft recipients: current concepts and future developments.
Liver Transpl 2016;22:1593–1602.
|
| 5 |
Leung DH, Narang A, Minard CG, Hiremath G, Goss JA, Shepherd R.
A 10-year united network for organ sharing review of mortality and risk factors in young children awaiting liver transplantation.
Liver Transpl 2016;22:1584–1592.
|
| 6 |
Manzoni D, D’Ercole C, Spotti A, Carrara B, Sonzogni V.
Congenital heart disease and pediatric liver transplantation: complications and outcome.
Pediatr Transplant 2007;11:876–881.
|
| 7 |
Concejero A, Chen CL, Liang CD, et al.
Living donor liver transplantation in children with congenital heart disease.
Transplantation 2007;84:484–489.
|
| 8 |
Feier FH, Neto JS, Porta G, et al.
Liver transplantation after stage II palliation for hypoplastic left heart syndrome.
Liver Transpl 2013;19:322–327.
|
| 9 |
Marino IR, ChapChap P, Esquivel CO, et al.
Liver transplantation for arteriohepatic dysplasia (Alagille's syndrome).
Transpl Int 1992;5:61–64.
|
| 10 |
Arguedas MR, Abrams GA, Krowka MJ, Fallon MB.
Prospective evaluation of outcomes and predictors of mortality in patients with hepatopulmonary syndrome undergoing liver transplantation.
Hepatology 2003;37:192–197.
|
| 11 |
Durand P, Baujard C, Grosse, AL, et al.
Reversal of hypoxia by inhaled nitric oxide in children with severe hepatopulmonary syndrome, type 1, during and after liver transplantation.
Transplantation 1998;65:437–439.
|
| 12 |
Pungpapong S, Kim WR, Poterucha JJ.
Natural history of hepatitis B virus infection: an update for clinicians.
Mayo Clin Proc 2007;82:967–975.
|
| 13 |
Ni YH, Huang LM, Chang MH, et al.
Two decades of universal hepatitis B vaccination in Taiwan: impact and implication for future strategies.
Gastroenterology 2007;132:1287–1293.
|
| 14 |
Chen HP, Tsai YF, Lin JR, Liu FC, Yu HP.
Recipient age and mortality risk after liver transplantation: a population-based cohort study.
PLoS One 2016;11:e0152324.
|
| 15 |
Chung PC, Chen HP, Lin JR, Liu FC, Yu HP.
Impact of preoperative chronic renal failure on liver transplantation: a population-based cohort study.
Ther Clin Risk Manag 2016;14;12:1855–1860.
|
| 16 |
Tessier ME, Harpavat S, Shepherd RW, et al.
Beyond the Pediatric end-stage liver disease system: solutions for infants with biliary atresia requiring liver transplant.
World J Gastroenterol 2014;20:11062–11068.
|
| 17 |
Sundaram SS, Mack CL, Feldman AG, Sokol RJ.
Biliary atresia: indications and timing of liver transplantation and optimization of pretransplant care.
Liver Transpl 2017;23:96–109.
|
| 18 |
Rauchfuss F, Zidan A, Scheuerlein H, Dittmar Y, Bauschke A, Settmacher U.
Waiting time, not donor-risk-index, is a major determinant for beneficial outcome after liver transplantation in high-MELD patients.
Ann Transplant 2013;18:243–247.
|
| 19 |
Siniscalchi A, Cucchetti A, Toccaceli L, et al.
Pretransplant model for end-stage liver disease score as a predictor of postoperative complications after liver transplantation.
Transplantat Proc 2009;41:1240–1242.
|
| 20 |
Lin HC, Xirasagar S, Chen CH, Hwang YT.
Physician’s case volume of intensive care unit pneumonia admissions and in-hospital mortality.
Am J Respir Crit Care Med 2008;177:989–994.
|
| 21 |
Wu YT, Lee HY.
National health insurance database in Taiwan: a resource or obstacle for health research?
Eur J Intern Med. 2016;31:e9–e10.
|
| 22 |
Liu PY, Cheng SB, Lin CC, et al.
Cytomegalovirus disease after liver transplantation: a nationwide population-based study.
Transplant Proc 2014;46:832–834.
|
| 23 |
Kaneko J, Sugawara Y, Tamura S, et al.
De novo malignancies after adult-to-adult living-donor liver transplantation with a malignancy surveillance program: comparison with a Japanese population-based study.
Transplantation 2013;95:1142–1147.
|
| 24 |
Chen HP, Tsai YF, Lin JR, Liu FC, Yu HP.
Incidence and outcomes of acute renal failure following liver transplantation.
Medicine 2015;94:e2320.
|